Therapeutic Targets for DOX-Induced Cardiomyopathy: Role of Apoptosis vs. Ferroptosis
Title: Therapeutic Targets for DOX-Induced Cardiomyopathy: Role of Apoptosis vs. Ferroptosis
Authors: Hiroki Kitakata, Jin Endo, Hidehiko Ikura, Hidenori Moriyama, Kohsuke Shirakawa, Yoshinori Katsumata, and Motoaki Sano
Journal: International Journal of Molecular Sciences, Vol: 23, Issue: 3 (2022)
DOI: 10.3390/ijms23031414
Abstract:
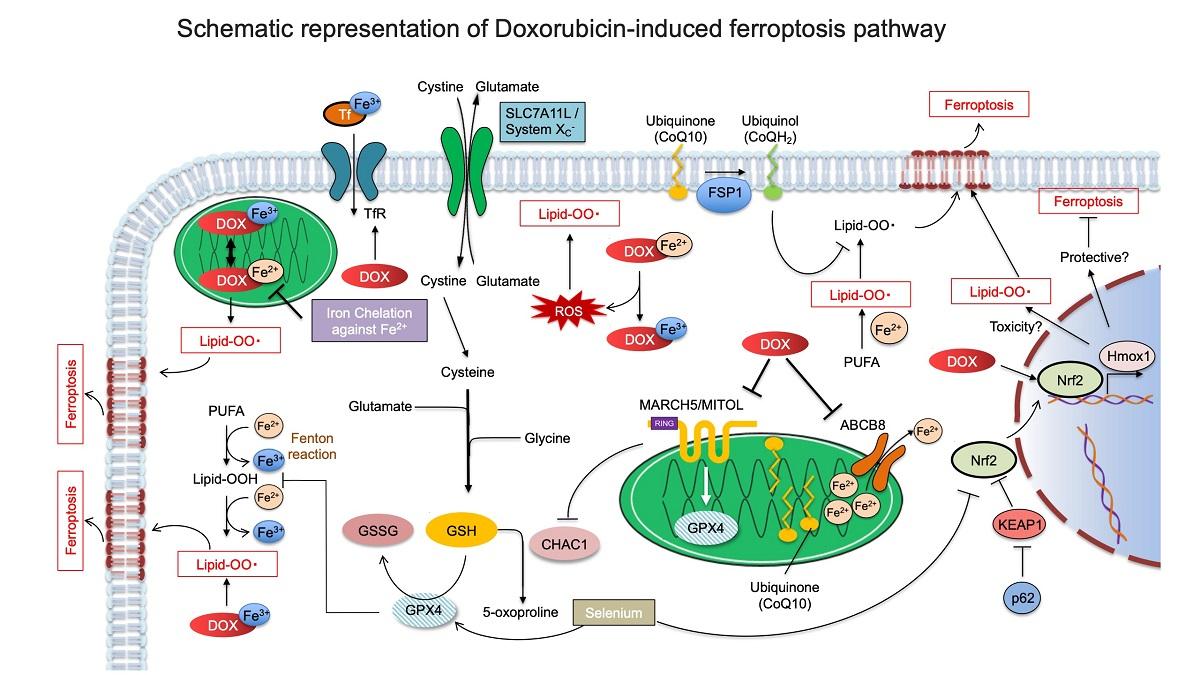
Doxorubicin (DOX) is the most widely used anthracycline anticancer agent; however, its cardiotoxicity limits its clinical efficacy. Numerous studies have elucidated the mechanisms underlying DOX-induced cardiotoxicity, wherein apoptosis has been reported as the most common final step leading to cardiomyocyte death. However, in the past two years, the involvement of ferroptosis, a novel programmed cell death, has been proposed. The purpose of this review is to summarize the historical background that led to each form of cell death, focusing on DOX-induced cardiotoxicity and the molecular mechanisms that trigger each form of cell death. Furthermore, based on this understanding, possible therapeutic strategies to prevent DOX cardiotoxicity are outlined. DNA damage, oxidative stress, intracellular signaling, transcription factors, epigenetic regulators, autophagy, and metabolic inflammation are important factors in the molecular mechanisms of DOX-induced cardiomyocyte apoptosis. Conversely, the accumulation of lipid peroxides, iron ion accumulation, and decreased expression of glutathione and glutathione peroxidase 4 are important in ferroptosis. In both cascades, the mitochondria are an important site of DOX cardiotoxicity. The last part of this review focuses on the significance of the disruption of mitochondrial homeostasis in DOX cardiotoxicity.
Related Articles:
- MITOL/MARCH5 determines the susceptibility of cardiomyocytes to doxorubicin-induced ferroptosis by regulating GSH homeostasis. Journal of Molecular and Cellular Cardiology, Vol. 161, pp. 116-129 (2021). DOI: 10.1016/j.yjmcc.2021.08.006.
- Imeglimin prevents heart failure with preserved ejection fraction by recovering the impaired unfolded protein response in mice subjected to cardiometabolic stress. Biochemical and Biophysical Research Communications, Vol. 572, pp. 185-190 (2021). DOI: 10.1016/j.bbrc.2021.07.090





