Scaffold-Free Tubular Engineered Heart Tissue From Human Induced Pluripotent Stem Cells Using Bio-3D Printing Technology in vivo
Title: Scaffold-Free Tubular Engineered Heart Tissue From Human Induced Pluripotent Stem Cells Using Bio-3D Printing Technology in vivo
Authors: Yujiro Kawai, Shugo Tohyama, Kenichi Arai, Tadashi Tamura, Yusuke Soma, Keiichi Fukuda, Hideyuki Shimizu, Koichi Nakayama, and Eiji Kobayashi
Journal: Frontiers in Cardiovascular Medicine, Vol: 8 (2022)
DOI: 10.3389/fcvm.2021.806215
Abstract:
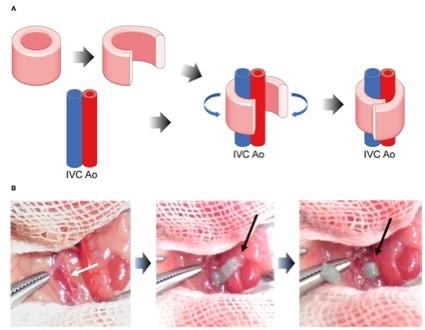
Engineered heart tissues (EHTs) that are fabricated using human induced pluripotent stem cells (hiPSCs) have been considered as potential cardiac tissue substitutes in case of heart failure. In the present study, we have created hiPSC-derived cardiac organoids (hiPSC-COs) comprised of hiPSC-derived cardiomyocytes, human umbilical vein endothelial cells, and human fibroblasts. To produce a beating conduit for patients suffering from congenital heart diseases, we constructed scaffold-free tubular EHTs (T-EHTs) using hiPSC-COs and bio-3D printing with needle arrays. The bio-3D printed T-EHTs were cut open and transplanted around the abdominal aorta as well as the inferior vena cava (IVC) of NOG mice. The transplanted T-EHTs were covered with the omentum, and the abdomen was closed after completion of the procedure. Additionally, to compare the functionality of hiPSC-COs with that of T-EHTs, we transplanted the former around the aorta and IVC as well as injecting them into the subcutaneous tissue on the back of the mice. After 1 m of the transplantation procedures, we observed the beating of the T-EHTs in the mice. In histological analysis, the T-EHTs showed clear striation of the myocardium and vascularization compared to hiPSC-COs transplanted around the aorta or in subcutaneous tissue. Based on these results, bio-3D-printed T-EHTs exhibited a better maturation in vivo as compared to the hiPSC-COs. Therefore, these beating T-EHTs may form conduits for congenital heart disease patients, and T-EHT transplantation can form a treatment option in such cases.
Related Articles:
- Efficient Large-Scale 2D Culture System for Human Induced Pluripotent Stem Cells and Differentiated Cardiomyocytes. Stem Cell Report, Vol. 9, Issue: 5, pp. 1406-1414 (2017). DOI: 10.1016/j.stemcr.2017.08.025.
- Fabrication of scaffold-free tubular cardiac constructs using a Bio-3D printer. PLOS ONE, Vol. 13, Issue 12 (2018). DOI: 10.1371/journal.pone.0209162
- Intramyocardial Transplantation of Human iPS Cell-Derived Cardiac Spheroids Improves Cardiac Function in Heart Failure Animals. JACC: Basic to Translational Science, Vol. 6, Issue: 3, pp. 239-254 (2021). DOI: 10.1016/j.jacbts.2020.11.017.





