Young age, female sex, and presence of systemic adverse reactions are associated with high post-vaccination antibody titer after two doses of BNT162b2 mRNA SARS-CoV-2 vaccination
Title: Young age, female sex, and presence of systemic adverse reactions are associated with high post-vaccination antibody titer after two doses of BNT162b2 mRNA SARS-CoV-2 vaccination: An observational study of 646 Japanese healthcare workers and university staff
Authors: Yoshifumi Uwamino, Toshinobu Kurafuji, Yasunori Sato, Yukari Tomita, Ayako Shibata, Akiko Tanabe, Yoko Yatabe, Masayo Noguchi, Tomoko Arai, Akemi Ohno, Hiromitsu Yokota, Wakako Yamasawa, Shunsuke Uno, Tomoyasu Nishimura, Naoki Hasegawa, Hideyuki Saya, Masatoshi Wakui, Mitsuru Murata, for the Keio Donner Project Team
Journal: Vaccine, Vol: 40, Issue: 7, pp. 1019-1025 (2022)
DOI: 10.1016/j.vaccine.2022.01.002
Abstract:
Background: SARS-CoV-2 vaccination has started worldwide, including Japan. Although high rates of vaccine response and adverse reactions of BNT162b2 vaccine have been reported, knowledge about the relationship between sex differences and antibody response is limited. Furthermore, it is uncertain whether adverse reactions are associated with the vaccine response.
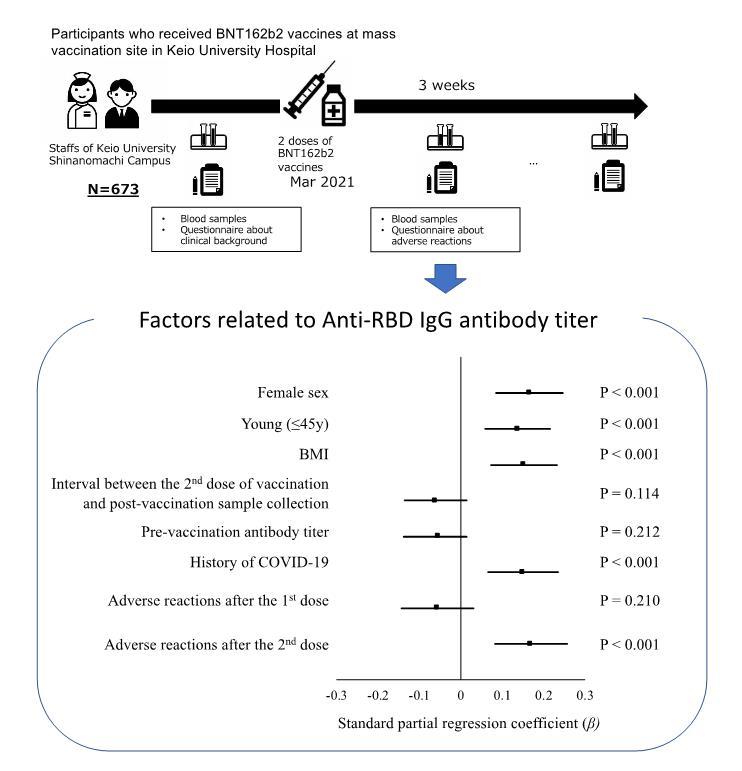
Methods: This prospective observational study included 673 Japanese participants working in a medical school and its affiliated hospital in Tokyo, Japan (UMIN000043340). Serum samples were collected before the first dose and three weeks after the second dose of BNT162b2 vaccine, and antibody titers against the receptor-binding domain of the spike protein of SARS-CoV-2 were measured. Answers to questionnaires about background characteristics and adverse reactions were obtained at the time of sample collection, and the relationship between antibody titers was analyzed.
Results: After excluding participants who did not complete receiving two doses of vaccination or two series of serum sample collection, 646 participants were analyzed. Although all participants became sero-positive after vaccination, antibody titers were highly variable among individuals (260.9-57,399.7A U/mL), with a median titer of 13478.0AU/mL. Mean titer was higher in females than in males and higher in young (≤45 years old) participants than in aged (>45 years old) participants. Participants who experienced adverse reactions demonstrated a higher antibody titer after vaccination than those without adverse reactions. Multivariable analysis demonstrated that young age, female sex, and adverse reactions after the second dose were independently related to higher antibody titers after the second dose.
Discussion: A favorable antibody response was observed after two doses of BNT162b2 vaccination among mostly healthy Japanese participants, especially among female and young participants. Although further investigation is essential, our results imply that the systemic adverse reactions (i.e., fever and general fatigue) are associated with a higher antibody response that indicates the acquisition of humoral immunity.
Related Articles:
- Assessing anti-SARS-CoV-2 cellular immunity in 571 vaccines by using an IFN-γ release assay. European Journal of Immunology, Vol. 52, Issue 12 (2022). DOI: 10.1002/eji.202249794.
- The effect of the E484K mutation of SARS-CoV-2 on the neutralizing activity of antibodies from BNT162b2 vaccinated individuals. Vaccine, Vol. 40, Issue 13, pp. 1928-1931 (2022). DOI: 10.1016/j.vaccine.2022.02.047
- Humoral and cellular immune response dynamics in Japanese healthcare workers up to six months after receiving a third dose of BNT162b2 monovalent vaccine. Vaccine, Vol. 41, Issue 9, pp. 1545-1549 (2023). DOI: 10.1016/j.vaccine.2023.01.049.





