Research Frontiers
"Metabolic domino and metabo-aging": Treatment of lifestyle-related diseases through studies on the metabolism of organs
Hiroshi Itoh, Professor
Department of Endocrinology, Metabolism and Nephrology, Keio University, School of Medicine.
December 25, 2017

Hiroshi Itoh, Professor, Department of Endocrinology,
Metabolism and Nephrology, Keio University School of Medicine.
Prof. Hiroshi Itoh and his colleagues focus on research and clinical activities regarding lifestyle-related diseases including obesity, diabetes, dyslipidemia, hypertension, and chronic kidney disease. "We are looking at these diseases from the viewpoint of molecular or energy metabolism to elucidate their pathology and develop treatments," says Itoh.
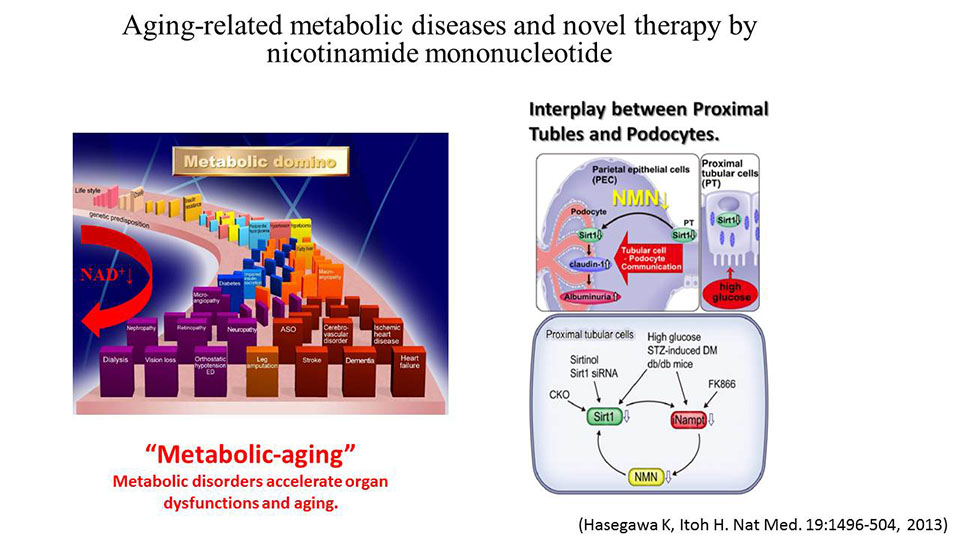
Important findings so far include the discovery that diseases such as diabetes and hypertension are not one independent disease, but accumulate due to the increase of visceral fat in an individual over time. Itoh refers to this concept as the "metabolic domino."
Since the metabolic domino is simply due to the process of aging, the KGRI researchers refer to such metabolic syndromes and aging associated with the metabolic domino as "metabo-aging." Importantly, a key substance of metabo-aging is NAD (nicotinamide adenine dinucleotide)--a sensor for energy metabolism. It is clear that the production of NAD in organs decreases due to aging, obesity, and diabetes. The researchers expect that preventing this reduction of NAD will lead to the prevention and treatment of metabo-aging.
Itoh and his collaborators discovered that diabetic kidney disease is triggered by a decrease in the activity of sirtuin (a molecule that causes longevity by calorie restriction), which leads to a decrease in the production of NMN (nicotinamide mononucleotide), a precursor of NAD. "Thus, we discovered that in the kidneys, metabo-aging was the cause of organ failure and we showed the possibility of treating chronic kidney disease using NMN," says Itoh.
Furthermore, in 2016, researchers at the Washington University in St. Louis reported that oral administration of NMN prevented age-related diseases such as obesity, diabetes, and osteoporosis in animal experiments. On the heels of this research, in July 2016, Itoh's group collaborated with Washington University to conduct the first studies in the world on the oral administration of NMN in humans.
After confirming the safety of NMN for humans, the researchers want to develop treatments using NMN for obesity, diabetes, and chronic kidney disease. Currently, they are investigating lifestyle-related diseases from the perspective of the metabolism of organs, and developing new treatment methods for these diseases. "I hope to continue this research in collaboration with people from all over the world," says Itoh.
Please contact the KGRI for further information about this research.
Contact
Keio University Global Research Institute (KGRI)
2-15-45 Mita, Minato-ku, Tokyo 108-8345 Japan
E-mail : kgri@info.keio.ac.jp
http://www.kgri.keio.ac.jp/en/index.html
Further information
Hiroshi Itoh, Professor
Department of Endocrinology, Metabolism and Nephrology, Keio University, School of Medicine.
https://keio.pure.elsevier.com/en/persons/hiroshi-itoh
Recent research
As of December 25, 2017
Insights into the molecular mechanisms leading to kidney dysfunction in diabetic patients
About one-third of diabetes patients suffer from renal failure. Therefore, understanding the mechanisms linking diabetes to renal damage (diabetic nephropathy) would benefit patients as it would help in developing new therapeutic targets and strategies. Sirtuin 1 (Sirt1) is a protein that is involved in cellular stress responses and has been implicated in diabetic nephropathy. However, the exact role of renal Sirt1 on the pathogenesis of renal damage in diabetes has not been fully elucidated.
read more >
Researchers in Japan have previously shown that Sirt1 overexpression can alleviate acute kidney injury in a Sirt1-overexpressing mouse model. The same group has now established the mechanism that links, at least in part, Sirt1 with the pathogenesis of renal damage in diabetes.
Shu Wakino and colleagues from Keio University, Shizuoka Red Cross Hospital and the Massachusetts Institute of Technology used Sirt1-overexpressing mice, Sirt1 knockout mice and diabetic mouse models to investigate the role of Sirt1 in protecting from diabetes-induced renal damage. Reduction of Sirt1 expression resulted in an increase in Claudin-1 levels and subsequently, albuminuria, which is an early marker of renal damage. The mechanism by which Sirt1 affects the levels of Claudin-1 was found to be through directly epigenetically regulating the expression of the Cldn1 gene. Furthermore, nicotinamide mononucleotide (NMN) was found to mediate the interplay between PTs and podocytes, which are important components of the kidney filtration apparatus, and hence would directly affect albuminuria. Using human renal biopsy samples, the group found that SIRT1 and Claudin-1 levels are also involved in diabetes-related albuminuria in humans.
Taken together the results demonstrate that "Sirt1 in PTs protects against albuminuria in diabetes by maintaining NMN concentrations around glomeruli, thus influencing podocyte function," the authors conclude. Furthermore, since observations in patient samples reflect some of the mouse model results the authors state that "the results of this study could contribute to new therapeutic strategies to prevent diabetes-induced albuminuria."
Publication and affiliations:
Authors: Kazuhiro Hasegawa1, Shu Wakino1, Petra Simic2, Yusuke Sakamaki3, Hitoshi Minakuchi1, Keiko Fujimura1, Kozi Hosoya1, Motoaki Komatsu1, Yuka Kaneko1, Takeshi Kanda1, Eiji Kubota3, Hirobumi Tokuyama1, Koichi Hayashi1, Leonard Guarente2 & Hiroshi Itoh1.
Title: Renal tubular Sirt1 attenuates diabetic albuminuria by epigenetically suppressing Claudin-1 overexpression in podocytes.
Journal: Nature Medicine 19 (2013)
DOI: 10.1038/nm.3363.
- Department of Internal Medicine, School of Medicine, Keio University, Tokyo, Japan.
- Department of Biology, Massachusetts Institute of Technology, Cambridge, Massachusetts, USA.,.
- 3. Department of Internal Medicine, Shizuoka Red Cross Hospital, Shizuoka, Japan
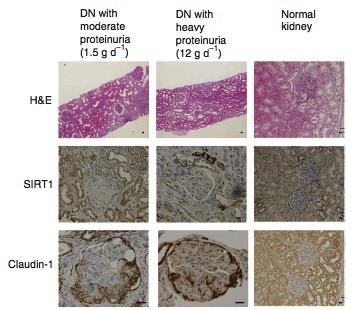
SIRT1 and Claudin-1 levels in kidney biopsies from patients with diabetic nephropathy (DN) and normal kidneys. The results show that SIRT1 levels in PTs and glomerular regions is lower, and Claudin-1 levels in the glomerular region is higher in patients with heavy protein urea compared to those with moderate protein urea.